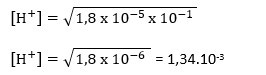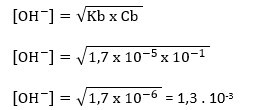Soal dan jawaban cara menentukan hasil perhitungan menentukan derajad keasaman, pH suatu larutan asam dan larutan basa. Soal ini berlaku pada larutan asam kuat dan basa kuat, juga asam dan basa lemah.
- Tentukan harga pH larutan 0,01 M HCl
Lihat Jawaban
HCl(aq) → H+(aq) + Cl–(aq)
[H+] = [HCl] = 0,001 M = 10-2 M
pH = -log [H+]
pH = -log 10-2
Jadi pH larutan = 2
- Berapa pH larutan 0,05 M H2SO4 ?
Lihat Jawaban
H2SO4(aq) → 2H+(aq) + SO42-(aq)
Dari persamaan reaksi
[H+] = 2 x [H2SO4]
[H+] = 2 x 0,005 = 0,1 M = 10-1 M
pH = -log [H+]
pH = -log 10-1
pH = 1
- Berapa [H+] dalam larutan HNO3 yang pHnya 2 ?
Lihat Jawaban
HNO3(aq) → H+(aq) + NO3-(aq)
pH = -log [H+]
2 = -log [H+]
log [H+] = -2
log [H+] = log 10-2
[H+] = 10-2 = 0,01 M
- Tentukan pH larutan 0,01 M NaOH !
Lihat Jawaban
NaOH(aq) → Na+(aq) + OH–(aq)
[OH–] = [NaOH]
[OH–] = 0,01 M = 10-2 M
pOH = -log [OH–]
pOH = -log 10-2
pOH = 2
pH = 14 – 2
pH = 12
- Berapa [OH–] yang terdapat dalam larutan KOH yang pHnya 13 ?
Lihat Jawaban
KOH(aq) → K+(aq) + OH–(aq)
pH = 13
pOH = 14 – 13 = 1
pOH = – log [OH–]
1 = – log [OH–]
log [OH–] = -1
log [OH–] = log 10-1
[OH–] = 10-1 = 0,1 M
- Tentukan pH larutan CH3COOH 0,1 M jika Ka CH3COOH = 1,8 x 10-5 !
Lihat Jawaban
Ca = [CH
3COOH] = 0,1 M = 10-1 M
[H
+] = √(Ka.Ca

pH = – log [H
+]
pH = – log 1,34.10
-3
pH = 3 – log 1,34
pH = 3 – 0,137
pH = 2,873
- Berapa pH larutan amonia 0,1 M yang tetapan ionisasinya 1,7.10-5 ?
Lihat Jawaban
NH
3(g) + H
2O(l) ⇄ NH
4+(aq) + OH
–(aq)
Cb = [NH
3] = 0,1 M = 10
-1 M

pOH = – log [OH
–]
pOH = – loh 1,3 . 10
-3
pOH = 3 – log 1,3
pOH = 3 – 0,114
pOH = 2,886
pH = 14 – pOH
pH = 14 – 2,886
pH = 11,114